|
7/23/2023 0 Comments Magnesium molar weight2003 (75) 683-800Ītomic weights of the elements 2011 by M.E. Explanation of how to find the molar mass of MgO: Magnesium oxide.A few things to consider when finding the molar mass for MgO:- make sure you have the corre. SOURCES Atomic weights of the elements: Review 2000 by John R de Laeter et al. However, the Commission does not recommend DSM3 as an international measurement standard for δ( 26/24Mg) measurements because it is not readily available to laboratories worldwide. Magnesium powder - Physico-chemical Properties Molecular Formula, Mg Molar Mass, 24.31 Density, 1.74 g/mL at 25 C (lit.) Melting Point, 648. Many of the δ( 26/24Mg) measurements reported herein were made using DSM3 as the standard. Understanding Magnesium (Mg) Magnesium is a chemical element with the symbol Mg and atomic number 12. Therefore, 6.02 x 1023 water molecules are in 18 grams of water. 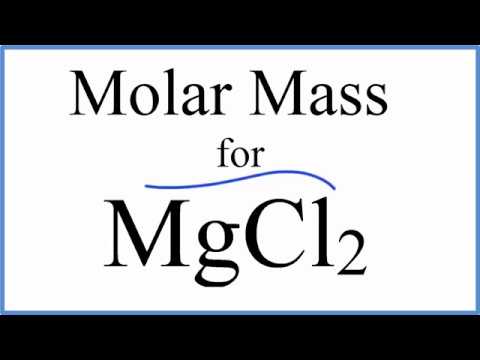
The standard atomic weight was determined by combining (1) the best calibrated isotope-ratio measurement of magnesium in DSM3 isotopic reference material (a mono-elemental nitric solution of magnesium),Īnd (2) the relative isotope-ratio differences between other magnesium-bearing materials and DSM3. Avogadro’s law states that 6.02 x 1023 molecules are in 1 mole of any substance. This change is intended to emphasize the fact that the atomic weight of magnesium is not a constant of nature, but depends upon the source of the material. The atomic weight, molecular weight, or formula weight of one mole of the fundamental units (atoms, molecules, or groups of atoms that correspond to the formula of a pure substance) is the ratio of its mass to 1/12 the mass of one mole of C 12 atoms, and being a ratio, is dimensionless. When the crucible is cool to touch, weigh the crucible and lid on the digital balance.In 2011, the Commission has changed the standard atomic weight of magnesium to A r(Mg) = based on an evaluation of the effect of variation in isotopic abundances in normal materials upon the atomic weight of magnesium. 
DO NOT put a hot crucible on the lab bench. Leave the crucible resting on the clay triangle. 
Turn off the burner and allow the crucible to cool. This will ensure that the crucible is clean and dry. Heat a clean and dry porcelain crucible with lid (on a clay triangle supported on a ring stand) directly over a Bunsen burner flame for about 5 minutes. The solid product from the reaction can be disposed into the regular garbage can in the lab. Let crucibles cool prior to adding water. It is important for the activity of many enzymes, especially those. Set hot crucibles on to wire screens to cool.Ī hot crucible will break if splashed with water directly. A metallic element that has the atomic symbol Mg, atomic number 12, and atomic weight 24.31. Use of eye protection is required for all experimental procedures.Ī hot crucible will break if placed directly on a cold surface. Section 2: Safety Precautions and Waste Disposalĭo not look directly at the burning magnesium ribbon. The flame is bright enough to damage your eye. Students will learn how to convert mass to mole of a given sample and determine empirical formula of a substance from mass and mole data. In this experiment, students will conduct the reaction between magnesium and oxygen gas. Students will determine the mass of magnesium sample before and after the reaction, and the mass of magnesium and oxygen in the product. Calculate the mole of a sample from its mass. In this experiment, students will conduct the reaction between magnesium and oxygen gas. \)ĭetermine the empirical formula of magnesium oxide.Ĭalculate the mass of oxygen using weighing-by-difference.Ĭalculate the mole of a sample from its mass.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
